2FIRSTS Exclusive Interview With FDA: Over 50,000 Inspections Have Been Carried Out in The Past 8 Months, And More Than Half Of Teenage Smokers Use ELFBAR Products
Leave a message
2FIRSTS exclusive interview with FDA: Over 50,000 inspections have been carried out in the past 8 months, and more than half of teenage smokers use ELFBAR products

Two Firsts (2FIRSTS) conducted an exclusive interview with Jim McKinney, the press director of the US FDA. Jim said that the FDA has conducted more than 50,000 compliance inspections on U.S. tobacco product retailers. In addition, in the United States, 50% of teenagers who use e-cigarettes choose the ELFBAR brand, while more than 20% of teenagers choose to use Esco Bar products.
FDA disclosed the core content:
1. In the past eight months, the FDA has conducted nearly 60,000 compliance inspections on U.S. tobacco product retailers and issued a total of 6,556 warning letters.
2.50% of teenagers who use e-cigarettes use ELFBAR products, and more than 20% use Esco Bar products.
3. Call on practitioners to "promptly and fully correct all violations and ensure compliance with all applicable laws and regulations."
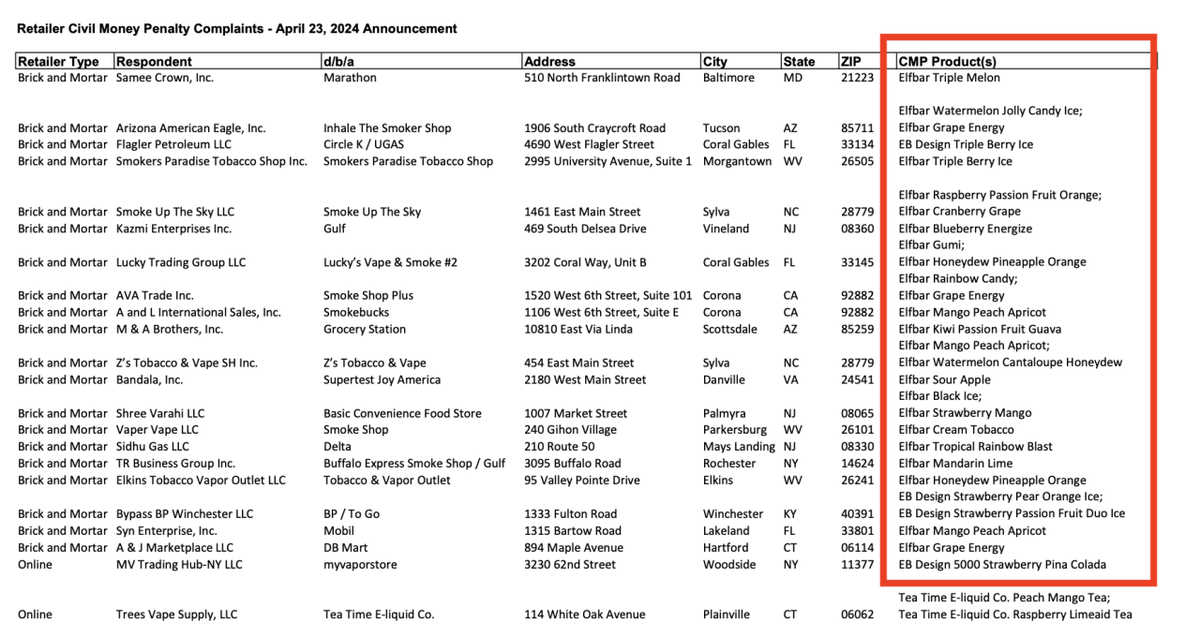
[Two Supremes are from Hong Kong, China] At the end of 2023, Reuters, AP, Times and other media conducted a concentrated report on the new tobacco industry; following this, the US political circles have paid more attention to the regulation of e-cigarettes in the near future. A wave of climax. The concept of "illegal e-cigarettes from China" has been mentioned many times in public. Public information shows that under compliance inspections of online and offline tobacco product retailers in the United States, the vast majority of civil fines were received in the batch on April 23. Most commercial entities are involved in the sale of the "ELFBAR" brand.
Moreover, after the United Kingdom and Australia implemented new e-cigarette restrictions, U.S. Senator Whip Dick Durbin wrote a letter at the end of January this year criticizing the FDA for its lack of enforcement, saying that the FDA had strict regulations on e-cigarette manufacturers' pre-market tobacco products. Review of product application (PMTA) "long-overdue" calls on FDA to take action to protect American children.

U.S. Senator Whip Dick Durbin criticized the FDA's public article|Source: dubin.senate.gov
How will pressure from the public opinion level be transmitted to regulatory policy formulation and implementation? Will FDA's compliance inspections become increasingly intensive and stringent? In response, the two supreme leaders visited the U.S. Food and Drug Administration (FDA). FDA Press Officer Jim McKinney accepted an exclusive interview.

Letter about Senator Durbin
Dubin stated on his personal website that the FDA's review of the premarket tobacco product application (PMTA) has been overdue for a long time. The deadline set by the federal court is September 9, 2021. As of January 2024, the FDA has missed the court regulations. has a deadline of 28 months and has requested action from the FDA on this issue.
Regarding the letter from Senator Durbin of the U.S. Senate, the FDA stated that it has indeed received the letter and will respond directly to the senator.
Warning letters against online retailers: Nearly 60,000 checks in eight months
As for the warning letters against online retailers, 2FIRSTS asked for details about compliance inspections of online and offline tobacco product retailers in the United States.
Jim pointed out in the interview that the FDA takes a comprehensive approach to tobacco product compliance and enforcement, including taking action against offenders throughout the supply chain, including manufacturers, importers, distributors and retailers, online Stores and offline physical stores are included in the scope of enforcement.
In the latest ongoing fiscal year (October 2023 to present), the FDA conducted a total of 57,191 inspections of commercial entities involved in tobacco business throughout the United States: 48,353 no violations were found, and a total of 6,556 warning letters and civil penalties were issued. Another 2,280 times a tobacco sales ban was issued.
The FDA issued the latest warning letter on May 1 to 14 online retailers selling unauthorized products: (For details, see: US FDA: More than 50% of teen smokers use ELFBAR e-cigarettes)
Brands involved: More than half reported using ELFBAR; Esco Bar accounts for more than 20% of the market
According to the latest FDA announcement on May 1 provided by Jim, the 2023 National Youth Tobacco Survey shows that among the more than 2.1 million American teenagers currently using e-cigarettes, more than 50% reported using Elf Bar products, and more than 20% reported using e-cigarettes. Weigh the use of Esco Bar products. A review of retail sales data and new internal data obtained from a survey of teenagers identified other brands included in these warning letters as being popular or appealing to teenagers.
As early as May 2023, the FDA issued an "Import Alert" (Import Alert 98-06). Most of the companies "marked in red" included manufacturers, transporters and distributors of ELFBAR, EBDESIGN, and Esco Bar brands. business.
Jim mentioned that after this alert, the FDA can effectively detain new tobacco products without marketing authorization without testing, and therefore called on practitioners to "timely and fully correct all violations and ensure compliance with all applicable laws and regulations."
Current enforcement results: More than 670 warning letters issued regarding new tobacco products
When asked about the current enforcement efforts, Jim cited FDA data and said that as of May 1, the FDA had issued orders to manufacturers, importers and distributors of illegally selling and/or distributing unauthorized new tobacco products (including e-cigarettes). issued more than 670 warning letters to manufacturers; issued more than 550 warning letters to retailers selling unlicensed tobacco products; and filed civil penalties against 55 manufacturers and more than 100 retailers for producing and/or selling unauthorized tobacco products. Litigation; cooperated with the U.S. Department of Justice (DOJ) to apply for injunctions against seven unauthorized e-cigarette product manufacturers, and cooperated with the DOJ to seize more than $700,000 worth of unauthorized e-cigarette products.
FDA: Issuing a warning letter first, which may later involve civil fines or a tobacco sales ban
When 2FIRSTS asked about the compliance inspection process, Jim responded that when the FDA first discovers a violation of the federal tobacco laws and regulations enforced by the FDA during a tobacco compliance inspection, it will notify traditional "brick and mortar" retail stores across the country as well as online retailers. and the manufacturer issued a warning letter.
Jim emphasized that failure to promptly and adequately correct all violations and ensure compliance with all applicable laws and regulations may result in FDA and other related enforcement actions, including civil fines or tobacco bans.
2FIRSTS will continue to pay attention to the US market and related legislation and law enforcement progress.






