US FDA issues warning letters to nine companies, Geek Bar and Lost Mary are named
Leave a message
US FDA issues warning letters to nine companies, Geek Bar and Lost Mary are named

The U.S. FDA issued warning letters to nine companies, accusing them of selling and distributing unauthorized flavored disposable e-cigarette products, including Geek Bar and Lost Mary, brands popular among young people.
On December 19, the U.S. Food and Drug Administration (FDA) official website announced that the FDA issued warning letters to eight online retailers and one manufacturer, accusing these companies of selling and/or distributing unauthorized flavored disposable e-cigarettes. Some of the unauthorized products mentioned in the warning letters used brand names popular among teenagers for marketing, including Geek Bar and Lost Mary. Other unauthorized products mentioned used the names or images of celebrities.
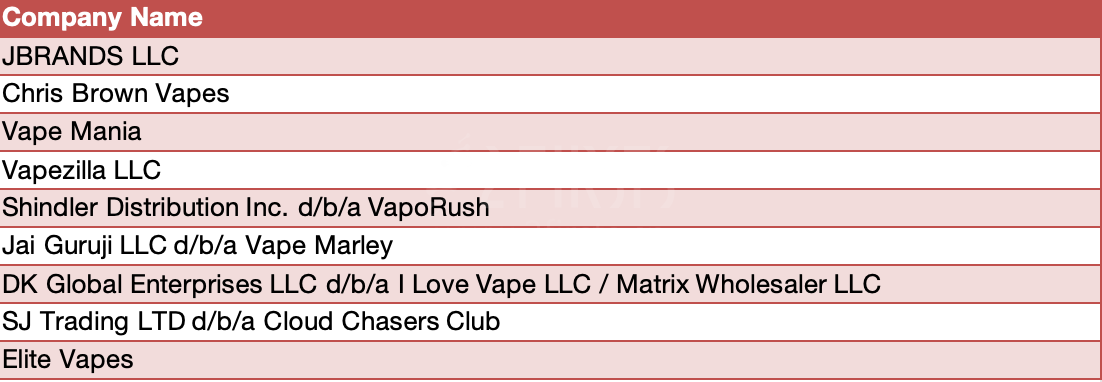
Warning letter list | Source: US FDA
The FDA said that the companies that received the warning letter sold or distributed products in the United States that have not been authorized by the FDA and therefore cannot be legally marketed in the United States, which violates the Federal Food, Drug, and Cosmetic Act. In addition to the violations noted in the letter, the companies were warned to address any violations that are the same or similar to those listed in the letter and to take necessary steps to comply with the law promptly. Failure to correct violations in a timely manner may result in other actions, such as bans, product seizures, and/or civil fines.
The FDA said it is committed to strict law enforcement and will continue to work with federal law enforcement partners when necessary to address the problem of unauthorized tobacco products in the United States. These latest warning letters demonstrate the FDA's continued efforts to remove unauthorized e-cigarette products from the market, especially those that attract young people. To date, the FDA has issued more than 700 warning letters accusing companies of manufacturing, selling and/or distributing unauthorized new tobacco products, issued more than 800 warning letters to retailers, and filed civil penalty lawsuits against 83 manufacturers and more than 175 retailers for the distribution or sale of unauthorized tobacco products.
As of December 19, 2024, the FDA has authorized 34 e-cigarette products and devices, and retailers can review the list of authorized products for sale.
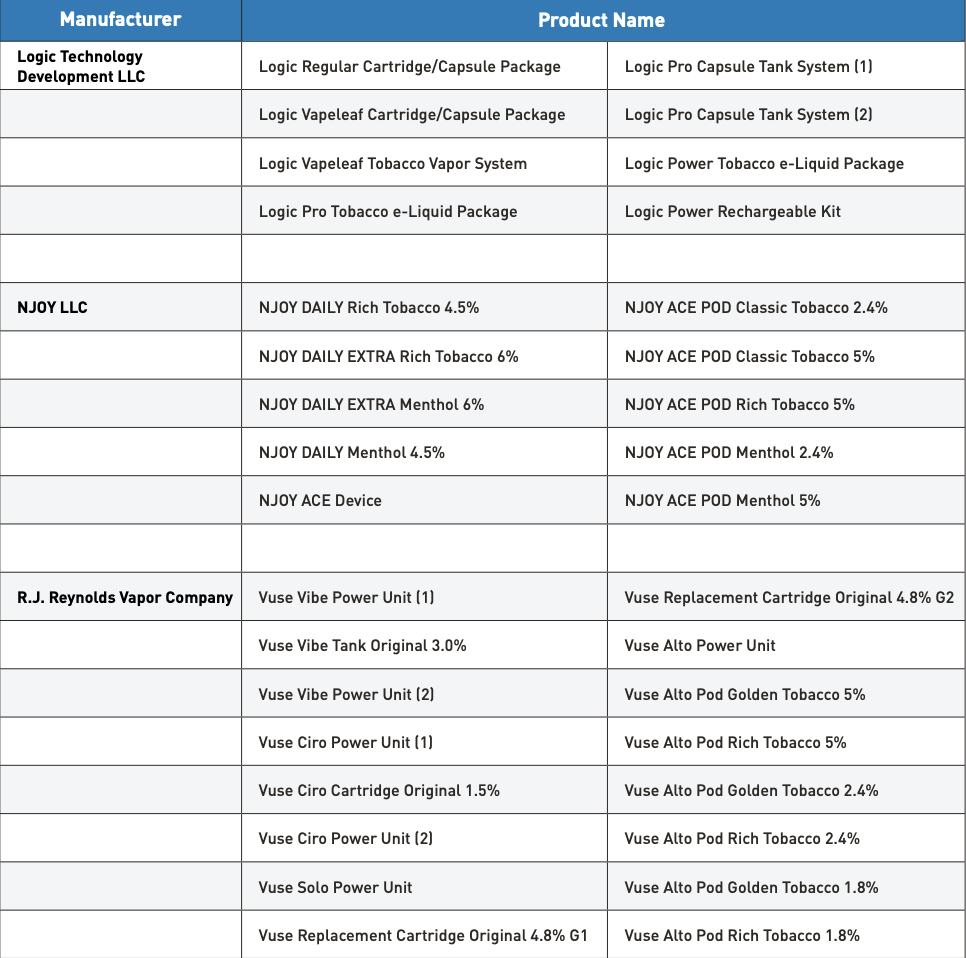
34 types of e-cigarette products | Source: US FDA









