US FDA issues warning letters to 115 businesses, Geek Bar and Elf Bar brands are named
Leave a message
US FDA issues warning letters to 115 businesses, Geek Bar and Elf Bar brands are named

The U.S. FDA and state partners issued warning letters to 115 retailers for selling unauthorized disposable e-cigarette products, including brands such as Geek Bar and Elf Bar. The merchants warned are required to submit a rectification plan within 15 working days, otherwise they will face further penalties.
On December 5, the U.S. Food and Drug Administration (FDA) announced that it had issued warning letters to 115 physical retailers who were punished for selling unauthorized e-cigarette products. These warning letters mentioned the sale of disposable e-cigarette products produced by Chinese manufacturers and sold under well-known brands, including Geek Bar Pulse, Geek Bar Skyview, Geek Bar Platinum and Elf Bar.
These warning letters are part of the FDA's ongoing enforcement actions coordinated by state partners to identify and combat the sale of unauthorized e-cigarettes. The FDA has contracts with states, regions or third-party agencies to assist in checking compliance at retail locations.
According to the results of the 2024 National Youth Tobacco Survey, 5.8% of current youth e-cigarette users reported using Geek Bar brand products. In addition, the FDA's review of additional rapid surveillance data and preliminary data from the Population Assessment of Tobacco and Health Study also confirmed that the brand has a high youth appeal.
The FDA closely monitors industry compliance with tobacco laws and regulations under the Federal Food, Drug, and Cosmetic Act and takes action when violations are found. Merchants who receive warning letters are required to respond within 15 business days and explain the corrective actions they will take and plans to prevent future violations. If the violations are not corrected in a timely manner, the FDA may take further action, including litigation, seizure and/or civil fines.
According to regulations, new tobacco products must be authorized by the FDA before they can be legally sold. Generally speaking, unauthorized products are subject to enforcement risks. To date, the FDA has authorized 34 e-cigarette products and devices. These are the only e-cigarette products that can currently be legally sold in the United States. For more information on tobacco products that can be legally sold in the United States, you can refer to the FDA's searchable tobacco product database.
The following is a specific list:

List of 115 physical retailers | Source: U.S. FDA
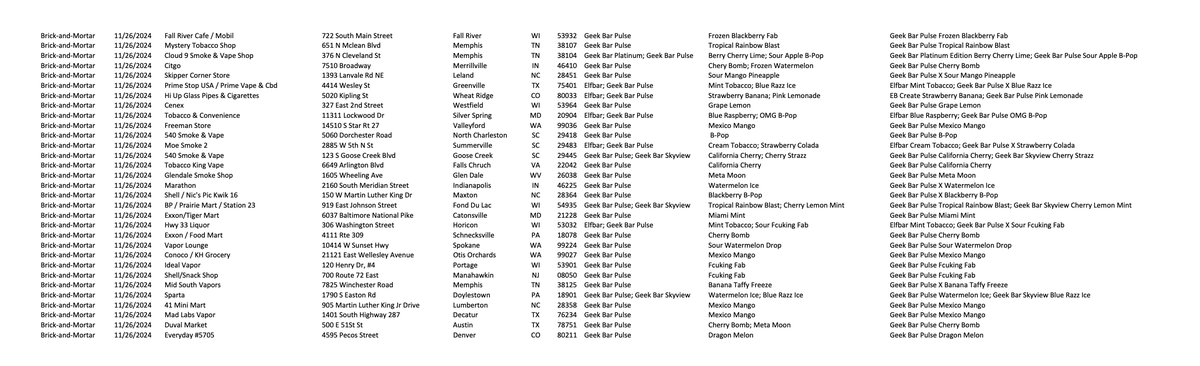
List of 115 physical retailers | Source: U.S. FDA









