The U.S. FDA has fined 18 retailers for selling unauthorized tobacco products
Leave a message
The U.S. FDA has fined 18 retailers for selling unauthorized tobacco products

The U.S. FDA announced that it has initiated fine procedures against two physical retailers and 16 online retailers. These retailers received warning letters for selling unauthorized tobacco products, but were found to have not taken corrective measures during subsequent inspections.
On November 26, the U.S. Food and Drug Administration (FDA) announced that the agency is seeking to impose fines on two physical retailers and 16 online retailers. The FDA had previously issued warning letters to these retailers, warning them to sell unauthorized tobacco products, but subsequent inspections found that these retailers failed to correct the violations.
The FDA said that failure to correct violations in a timely manner may result in additional enforcement actions, such as civil fines. The FDA said it is always committed to addressing the problem of unauthorized tobacco products throughout the supply chain. To date, the FDA has filed civil penalty complaints against 79 manufacturers and 175 retailers, accusing them of distributing or selling unauthorized tobacco products. In addition, the FDA works closely with federal law enforcement partners, including through the newly announced federal interdepartmental working group. For example, the FDA and the U.S. Customs and Border Protection recently announced the seizure of $76 million worth of illegal e-cigarettes.
As of press time, the FDA has approved 34 e-cigarette products and devices, and the agency maintains a printable one-page flyer listing all authorized e-cigarette products that retailers can easily review to determine which products can legally be sold in the U.S. Entities that manufacture, import, sell, or distribute e-cigarettes may be at risk of enforcement action if they lack the required premarket authorization.
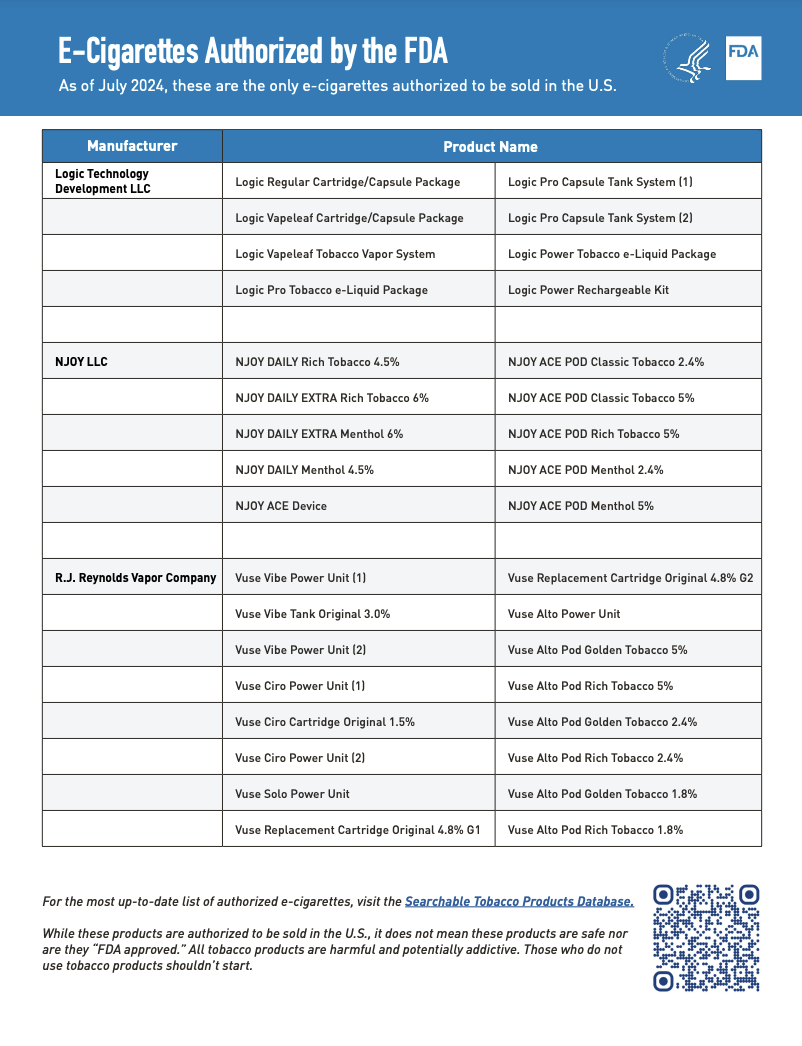
34 compliant e-cigarette products and devices | Source: FDA









