Many well-known brands are promoting 0-nicotine e-cigarettes in the United States. Does the FDA have no authority to regulate them?
Leave a message
Many well-known brands are promoting 0-nicotine e-cigarettes in the United States. Does the FDA have no authority to regulate them?

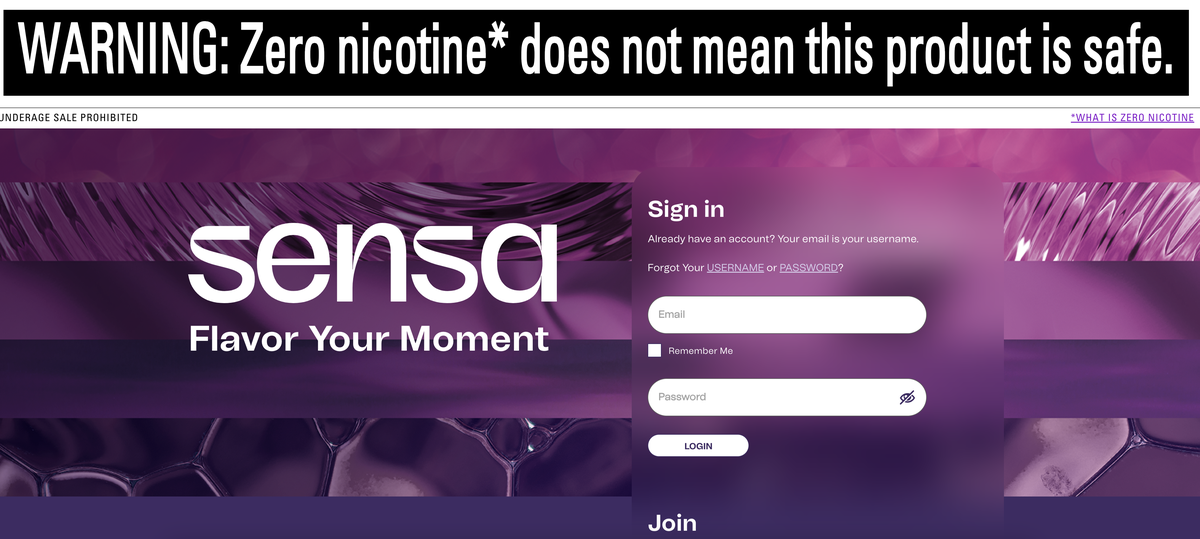
With Reynolds Tobacco launching the 0 nicotine e-cigarette brand Sensa in the United States, e-cigarette brands such as Geekbar and RAZ have begun to update and launch 0 nicotine versions of e-cigarette products. Are 0 nicotine products regulated by the FDA? Why do consumers choose 0 nicotine products?
Recently, with Reynolds Tobacco, a subsidiary of British American Tobacco (BAT), launching the 0 nicotine e-cigarette brand Sensa in the United States, the two tops found that more e-cigarette brands have begun to update and launch 0 nicotine versions of e-cigarette products in this market.
Demand Vape, an online e-cigarette wholesale store in the United States, found that e-cigarette brands including GeekBar and Raz have recently launched 0 nicotine versions of e-cigarettes. The appearance and configuration of these products are basically the same as the same model, except that the nicotine oil concentration of the product is reduced to 0% in the nicotine oil composition.

These changes are reflected in the appearance of the product. The most direct and impactful one is the front of the outer packaging, which is printed with "ZERO NICOTINE" in bold font.
Renault: No plans to seek federal approval for 0 nicotine e-cigarette products
Previously, Renault announced the launch of the 0 nicotine e-cigarette brand Sensa, which went on sale on July 1 and offers six flavors, including watermelon frost, passion fruit frost and berry fusion. It can provide 5,000 puffs and comes with a recyclable battery, with a retail price of $19.99.
This brand is not BAT's only product in the zero-nicotine field. In Japan, BAT announced that it would launch its new nicotine-free disposable e-cigarette product "Vuse Zero Nic Sensations" globally on the same day.
In a press release, BAT said that the product belongs to the e-cigarette brand "Vuse". Sensations has launched four flavors and is sold in FamilyMart convenience stores, Amazon and vape studio platforms in Tokyo.

Comparing Sensa and Vuse Zero Nic Sensations under British American Tobacco Group, the two are identical in design and positioning. The word Sensations is simplified to Sensa and upgraded to a separate brand.
At present, Vuse has a total of four products that have passed PMTA in the United States-Vuse Alto, Vuse Vibe, Vuse Ciro and Vuse Solo.
Sensa's sales in the United States avoid Vuse. It has a separate e-cigarette website sensavape to distinguish it from Vuse's sales website vusevapor. Both websites are marked with ©2024 R.J. Reynolds Vapor Company, which comes from the same parent company.
Deliberately distinguishing development in the US market may help avoid direct competition between brands, ensure that its products have their own unique position in the market, and provide customized choices for different consumer groups. It specifically chose to avoid brand confusion with Vuse in the US region and used its positioning strategy to attract consumers with different needs.
It should be noted that in the United States, where the regulation of zero-nicotine atomization products is still in a gray area, it also leaves room for the FDA and other regulatory agencies to take action.

In an interview with the Wall Street Journal, Reynolds said it does not plan to seek federal approval for Sensa. Because these devices do not contain nicotine and are not compatible with any other tobacco products, they are not under the jurisdiction of the FDA's Center for Tobacco Products (CTP).
In response to relevant questions, the FDA declined to specify whether nicotine-free e-cigarette products like Sensa fall under its regulatory scope.
FDA's regulatory loopholes
Currently, according to the FDA's latest version of the "E-Cigarettes, Vapes, and other Electronic Nicotine Delivery Systems (ENDS)Subscribe to Email Updates" document, its definition of e-cigarette products: e-cigarettes, vaporizers, e-cigarette pens, hookah pens, electronic cigarettes (e-cigarettes or e-cigs), electronic cigars and electronic pipes are tobacco products of electronic nicotine delivery systems (ENDS).
They Use "e-liquid" (industry-speak for "smoke oil"), which usually contains nicotine from tobacco as well as flavorings, propylene glycol, vegetable glycerin and other ingredients. The liquid is heated to produce an inhaled aerosol.
In the article "How FDA is Regulating E-Cigarettes Subscribe to Email Updates", FDA Acting Commissioner Ned Sharpless conveyed to the outside world the main responsibilities of CTPs responsible for regulating tobacco products and how to regulate e-cigarette products. He mentioned that the FDA requires all ENDS manufacturers on the market to meet the following requirements, including but not limited to:
Registering institutions and submitting product lists, including labels and advertisements
Submitting tobacco health documents
Submitting ingredient lists
Including mandatory warning statements on the packaging and advertisements of ENDS (including certain ENDS ingredients, such as e-liquids), the statement reads: "Warning: This product contains nicotine. Nicotine is an addictive chemical."
It is worth noting that according to the previous U.S. Tobacco Control Act, synthetic nicotine was not included in the regulation. The FDA defines nicotine only as the nicotine substance of natural tobacco. In order to prevent e-cigarette companies from bypassing the review of products containing synthetic nicotine, a new law passed in the United States in April 2022 clarified the FDA The power to regulate tobacco products containing nicotine from any source, including synthetic nicotine.

The law states that, starting April 14, 2022, manufacturers, distributors, importers, and retailers of tobacco products containing non-tobacco nicotine (NTN) (i.e., nicotine that is not made or derived from tobacco, such as synthetic nicotine) must ensure compliance with applicable requirements under the Federal Food, Drug, and Cosmetic Act (FD&C Act) resulting from this law, such as:
Not sell these products to persons under 21 years of age (whether in person or online);
Not market these products as modified risk tobacco products without FDA authorization; and
Not distribute free samples of these products.
In addition, owners and operators of facilities that manufacture, prepare, compound, or process NTN products must register with the FDA and list all tobacco products they manufacture, prepare, compound, or process for commercial distribution.
But if e-cigarettes themselves do not contain any nicotine cost, does the FDA have regulatory authority over this?
At present, the FDA's regulatory authority has not been specifically clarified. In addition to the FDA's "refusal to explain" Sensa's nicotine products, there is also a case in 2017 that best confirms this statement.
In 2017, in the case of "Nicopure Labs, LLC v. U.S. Food and Drug Administration", Nicopure Labs challenged the "determination rule" of the "Family Smoking Prevention and Tobacco Control Act of 2009" (TCA), which applies to electronic nicotine delivery system (ENDS) products, including e-cigarettes and e-liquids. Nicopure Labs believes that the decision exceeds the FDA's authority and that the FDA's review is "arbitrary and capricious."

Ultimately, the court upheld the FDA's decision to regulate e-cigarettes as "tobacco products" under the Tobacco Control Act. The court determined that the FDA acted within its authority and that it was reasonable to require e-cigarette manufacturers to undergo pre-market review and comply with marketing restrictions, and that it did not infringe constitutional rights.
But it is worth noting that the court determined in its ruling that in the FDA's "determination rule," the FDA defines "components or parts" as "any combination of software or materials that is designed or reasonably expected to:
(1) change or affect the performance, ingredients, components, or properties of tobacco products;
or (2) be used with tobacco products or for human consumption."
The important premise is that "they change or affect the performance of tobacco products and are used with tobacco products." Closed e-cigarettes, such as closed cartridges or disposable e-cigarettes, obviously cannot change "performance, ingredients, components, or properties" by adding e-liquid, nor can they be "used with tobacco products or for human consumption."
This is why Reno dared to claim in the interview: These devices are completely independent and incompatible with any tobacco products, so there is no need to seek FDA approval.
Due to the FDA's ambiguous attitude, it also indirectly affects the local state's regulation of such e-cigarettes. Take Louisiana as an example. The state's Office of Alcohol and Tobacco Control has published a "white list" called V.A.P.E. on its official website, listing all e-cigarettes or alternative nicotine products approved by the U.S. Food and Drug Administration (FDA). Since the bill came into effect, all e-cigarette products in Louisiana that have not been approved by the FDA cannot be legally sold, and merchants need to remove products not included in the list.
Given that the FDA's jurisdiction is unclear, zero-nicotine products are still in a "gray area". Whether it is the central federal government or local states, regulatory agencies that follow the FDA's provisions have not placed them under the same regulation as nicotine-containing (including synthetic nicotine).
Why do consumers choose zero-nicotine products?
Nicotine is addictive and is also the main reason for people's addiction to tobacco. Whether it is nicotine extracted from natural tobacco leaves or artificially synthesized nicotine, their basic chemical structure and addictive effect are the same.
Vaping e-cigarettes were originally designed as a tool to help smokers quit smoking. The principle is to gradually reduce smokers' dependence on traditional tobacco by providing nicotine. E-cigarettes allow users to meet their nicotine needs by inhaling nicotine-containing aerosols, while avoiding the harmful substances produced when traditional tobacco is burned.
However, over time, some e-cigarette users may choose to reduce the concentration of nicotine in the e-cigarettes they use to reduce their dependence on nicotine. This process is called "nicotine laddering", and users may eventually stop using nicotine-containing e-cigarettes completely or switch to 0-nicotine e-cigarette products by gradually reducing nicotine intake.
This part of users can continue to enjoy the sensory experience of e-cigarettes, such as the throat hit and taste of the aerosol, while avoiding the addictiveness of nicotine, just maintaining the physiological behavior of smoking.
On Reddit, a social platform known as the "American Post Bar," many e-cigarette users are looking for zero-nicotine e-cigarette products on the market. Many of them are considering temporarily giving up smoking for their families. "When I can't smoke medical marijuana (MMJ) in front of my family, I will use it."

Some e-cigarette consumers like the various flavors provided by e-cigarettes, do not want the effects of nicotine, and prefer the sensory experience of e-cigarettes.
"My wife smoked, but in an effort to quit, she switched to a vape. She had a nicotine e-cigarette. … I took a few puffs of her nicotine e-cigarette and it felt good, but I didn't want to smoke and didn't want to deal with the potential of addiction. So, we bought a few boxes of zero-nicotine e-cigarettes from a local tobacco shop."

On the other hand, some people use zero-nicotine e-cigarettes out of social needs, as the American Lung Association pointed out in an article, "Peer pressure - their friends encourage them to try smoking and continue smoking."
This kind of social need has led some people to use zero-nicotine e-cigarettes, which allows them to participate in social activities without taking nicotine.







