Interpretation | Nicotine pouches are less attractive to non-smokers: Research by the American behavioral research organization ARAC under the FDA compliance framework
Leave a message
Interpretation | Nicotine pouches are less attractive to non-smokers: Research by the American behavioral research organization ARAC under the FDA compliance framework

ARAC, an American behavioral research organization, presented a new study at the CORESTA 2024 conference. The study was conducted within the PMTA framework under FDA supervision and showed that nicotine pouches were less attractive to non-smokers regardless of flavor, which further supports the feasibility of oral nicotine. 2Firsts explores how these findings affect the future of the industry in the current chaotic situation of the nicotine pouch market, including competition from compliant tobacco companies and e-cigarette players, as well as the emergence of gray markets and counterfeit products, which have driven the need for greater compliance.
Author: Echo Guo
Co-founder & COO of 2Firsts
Editor's Note
At the 2024 CORESTA conference, ARAC Chief Research Officer Dr. Jessica Zdinak presented the company's latest research results. As a leading global media and consulting company in the new tobacco products (NGP) industry, 2Firsts was very interested in these research results and worked with ARAC to obtain the presentation PPT. This article, written by 2Firsts, is based on these PPTs and key insights from the ARAC study, combined with current industry trends, to explore the study's impact on nicotine pouches, their potential impact on public health, and changes in the regulatory framework.
Introduction
In the rapidly developing new tobacco product market, oral nicotine pouches are seen as a promising solution to reduce health risks. According to the latest insights from Verified Industry, the global oral nicotine pouch market is expected to soar from $1.2 billion in 2023 to $5.5 billion in 2033. Major tobacco companies, including Philip Morris International (PMI) and Altria, have already dominated this rapidly expanding market. At the same time, e-cigarette companies, especially those with supply chains based in China, are rapidly entering the nicotine pouch field, trying to seize this growth opportunity and increase competition in the field.
Currently, the United States is the largest nicotine pouch market and a barometer of the global market. Despite increasing public attention to the potential of nicotine pouches in reducing health risks and convenience, their appeal to non-tobacco users, especially teenagers, has caused increasing concerns. According to the FDA's CTP (Center for Tobacco Products), this category also requires companies to conduct a Tobacco Product Perception and Intention (TPPI) study during the new tobacco product application (PMTA) process.
At the CORESTA 2024 conference, ARAC (American Research Agency) presented a key TPPI study on public perception of oral nicotine white pouches (ONWP). The study showed that non-users had lower intentions to try or use ONWP compared to smokers.
Three key findings of the study: arguments in favor of oral nicotine white pouches
1. Intention comparison between users and non-users:
A key conclusion of the study was the comparison of intention to try ONWP between users and non-users. Non-users had significantly lower intentions to try flavored and tobacco-flavored ONWP than users. An important aspect of the study also explored whether flavors or tobacco flavors were more attractive to non-users. The findings showed that for non-users, the intention to try or use ONWP was similar between flavored and tobacco-flavored products.
2. Relative risk perception:
Both users and non-users perceived similar health risks for the entire nicotine pouch category, including flavored ONWP and tobacco-flavored ONWP. Most participants believed that the perceived health risks of ONWPs were lower than those of cigarettes, while others believed that the risks of using ONWPs were comparable to those of smoking. This suggests that ONWPs were evaluated more positively in terms of risk perception.
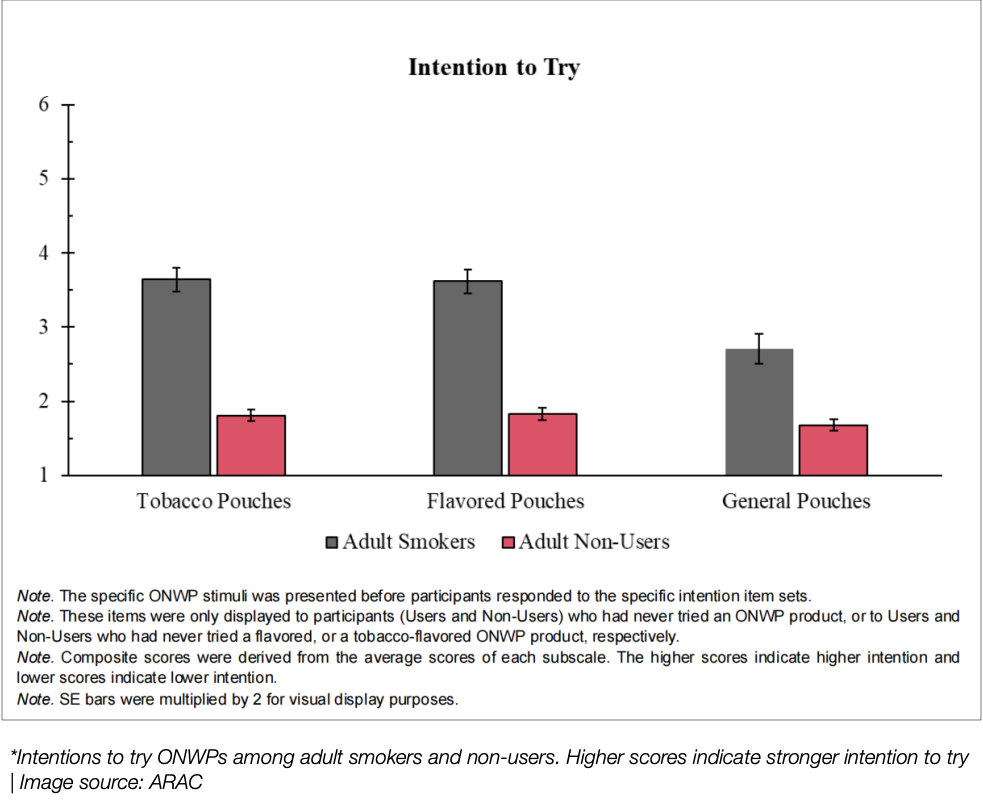
Intentions of adult smokers and non-users to try ONWPs, with higher scores indicating a stronger willingness to try | Source: ARAC
3. Reasons for using ONWPs:
The survey showed that the main attraction was the perceived lower harm compared to smoking. Both users and non-users agreed that "using ONWPs is less harmful to me than smoking" was the top reason for use. While the top reasons for using ONWPs varied between groups, this shared perception of reduced harm was always central.
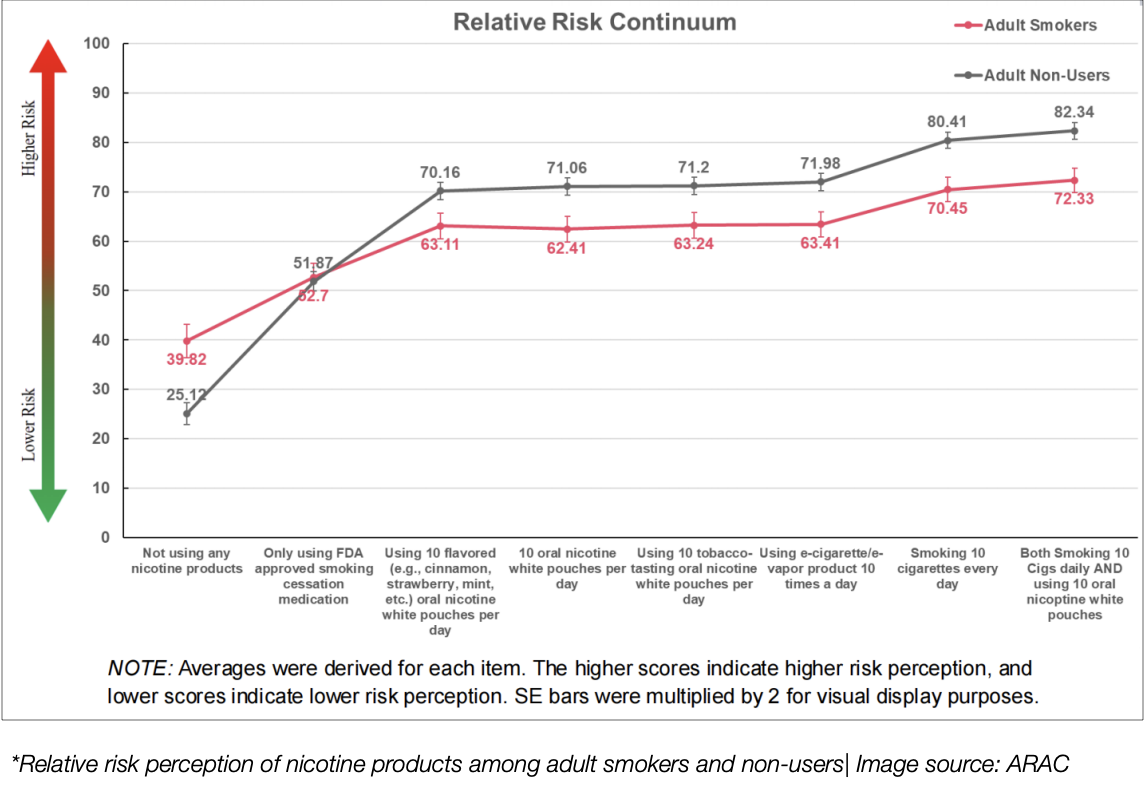
Relative risk perception of nicotine products among adult smokers and non-users | Source: ARAC
How ARAC conducts nicotine pouch TPPI under FDA compliance
The study was conducted between October and November 2023 and involved 1,423 American adults, including 432 smokers and 991 non-smokers. Participants were recruited through an online screening process to ensure that specific inclusion criteria were met, and the study focused on adults aged 21 to 65 years.

ONWP TPPI Research Group | Image source: ARAC
The study tested three types of oral nicotine white pouches (ONWP): plain, flavored, and tobacco-flavored. Participants were asked about their intention to try, intention to use, or likelihood to switch to the product, and their perception of associated risks after viewing the product stimulus.
The study design explored two key dimensions:
• Comparison of tobacco users and non-users: The study examined how tobacco users and non-users viewed ONWP products, focusing on their intention to try and use the product and their perception of risk.
• Intention and risk attitude: The study assessed participants' behavioral intentions (whether they would try or continue to use ONWP) and their attitudes toward perceived risks of using these products.
Research Impact: Dissecting Misconceptions, Informing FDA Policy, and Guiding Industry Innovation
ARAC's TPPI study findings represent a critical moment, especially in terms of the appeal of nicotine pouches to non-smokers. By addressing long-standing misconceptions, this study provides a clearer understanding of the role of these products in harm reduction and public health.
The study provides key insights for regulatory decision-making, showing that ONWPs can significantly reduce health risks for smokers while posing minimal risk to non-smokers. The findings meet the FDA's "appropriateness for the protection of public health" (APPH) standard, indicating that ONWPs may meet the criteria for PMTA approval.
The study also showed that the nicotine pouches were consistent with FDA standards, suggesting that the compliance process may be accelerated. This will reduce gray market and counterfeit products and create a more transparent, regulated market. Companies committed to compliance will benefit from this. In addition, the study's finding that flavored nicotine pouches are less attractive to non-smokers than tobacco flavors may spur innovation in flavor development, provided that it is scientifically validated and meets regulatory standards.
Study Limitations: Lack of Youth Data
A limitation of this study is that it focused only on adult participants, ignoring a key population: adolescents. Flavors in tobacco products are widely believed to attract young users. However, the study's conclusions - indicating that flavors are less attractive to non-smokers - are based only on adult data. This constitutes a significant gap, as adolescents may respond differently to flavored products due to social and developmental factors.








