Analysis of FDA Regulatory Science Policy Memorandum: Age Verification Products Receive Priority Review, Non-Tobacco Flavors Need to Prove Incremental Benefit
Leave a message
Analysis of FDA Regulatory Science Policy Memorandum: Age Verification Products Receive Priority Review, Non-Tobacco Flavors Need to Prove "Incremental Benefit"

On November 21, the FDA issued 13 scientific policy memoranda on PMTA review. 2Firsts communicated with several compliance experts and found that e-cigarette products with "age verification technology" will be reviewed first; at the same time, the FDA disclosed that non-tobacco flavored e-cigarettes need to prove "incremental benefits" for adult smokers, and applications that fail to provide randomized controlled trials or long-term research data may be considered "fatal flaws."
On November 21, the U.S. Food and Drug Administration (FDA) issued 13 memoranda on scientific policy for the review of pre-market applications for e-cigarettes, spanning 2020 to 2023. The FDA said these memoranda describe the process and priority methods used to file and review flavored e-cigarettes and other PMTAs (details: U.S. FDA issues 13 regulatory science policy memoranda: Focus on flavored e-cigarette review methods).

FDA releases PMTA-related regulatory science policy memorandum | Source: FDA
Regarding the content of the memorandum released this time, 2Firsts consulted relevant materials and communicated with many compliance experts and found that the importance of "age verification technology" was clearly stated, that is, devices with age verification will receive priority review. At the same time, some memoranda also disclosed the details of the FDA's additional requirements for flavor research in later reviews.
Products with age verification can receive priority review
A memorandum titled "Filing Prioritization for PMTAs received between September 10, 2020 to November 3, 2021" details the FDA's policy and background for prioritizing received pre-market tobacco product applications (PMTAs).

Filing Prioritization for PMTAs received between September 10, 2020 to November 3, 2021 Memorandum | Source: FDA
In the memorandum, the FDA explained the core principles of its review priorities, which mainly include the following two aspects:
Based on submission date: PMTAs are usually reviewed in the order of submission dates. For example, products submitted on September 10 will be prioritized over applications submitted on September 15.
Special priorities: Some applications with special technologies or public health significance may be prioritized. For example, the memorandum specifically mentioned that a company submitted a PMTA with "age-gating technology", which was prioritized due to its potential public health impact. The FDA stated that "the application was prioritized for Filing because it contains purported age-gating technology."

"The application was prioritized because it contains the alleged age verification technology | Source: FDA
"The memorandum shows the fact that products with "age verification" can be reviewed faster and passed the second round of review, directly entering the third stage of review." said Kurt, a compliance expert consultant at 2Firsts.
Kurt said, "My understanding is that the FDA will prioritize the review of age verification products and directly accelerate the queue to the third stage of review, but the review standards have not been lowered. "
He also believes that the FDA's review logic for e-cigarettes can be seen from this: devices with age verification functions can effectively prevent teenagers from using them, so they are considered a priority.
He said that such products may not need to undergo expensive randomized controlled trials (RCTs) or long-term cohort studies (Longitudinal Cohort Studies), because their terminal devices have already played a preventive role in adolescent abuse.
Flavor products need to prove "incremental benefits"
In another memorandum titled "ENDS Containing Non-Tobacco Flavored E-Liquid: Approach to PMTAs1 not in Substantive Scientific Review (Phase III)", the FDA discussed the pre-market tobacco product application (PMTA) management strategy for non-tobacco flavored e-cigarettes (ENDS).
In the memorandum, the FDA explained its own review standards. The agency cited Section 910 of the U.S. Federal Food, Drug, and Cosmetic Act (FD&C Act). Whether the product is suitable for "protecting public health" (APPH) is the key to the review. Non-tobacco flavored e-cigarettes need to prove that they have "incremental benefits" (incremental benefits) for adult smokers compared to tobacco flavored products. benefit).
"Incremental benefit" can be understood as the FDA requiring non-tobacco flavor e-cigarette submitters to prove that they provide additional benefits to adult smokers compared to tobacco flavor products or other existing options, and the evaluation requires evidence from randomized controlled trials (RCTs) or long-term cohort studies.
If the application lacks the above research, it will be regarded as a "fatal flaw" and may result in a marketing denial order (MDO).

Description of "fatal flaw" | Source: FDA

Description of "fatal flaw" | Source: FDA
Kurt said that since July 2021, the FDA has added long-term clinical testing requirements for flavors to the review process. This additional requirement has caused a large number of companies to be judged unqualified, and thousands of market rejection orders have been issued to companies. Subsequently, these companies have filed multiple lawsuits against the FDA, arguing that this additional requirement violates the original review standards of PMTA.
So how does the FDA define "non-tobacco flavors"?
A memorandum titled "Addendum to Approach to PMTAs1 for Non-Tobacco Flavored ENDS2 not in Substantive Scientific Review (Phase III)" provides an answer. The FDA clearly lists which products belong to non-tobacco flavors in this sub-memorandum and defines the scope of "tobacco" and "mint" flavors.
Kurt said that the tobacco flavors and menthol flavors in these two lists will not be reviewed as flavor products. Only FLAVOR SKU will be subject to fatal flaw long-term clinical trial review. If there is no relevant evidence, the FDA will send them an MDO letter.
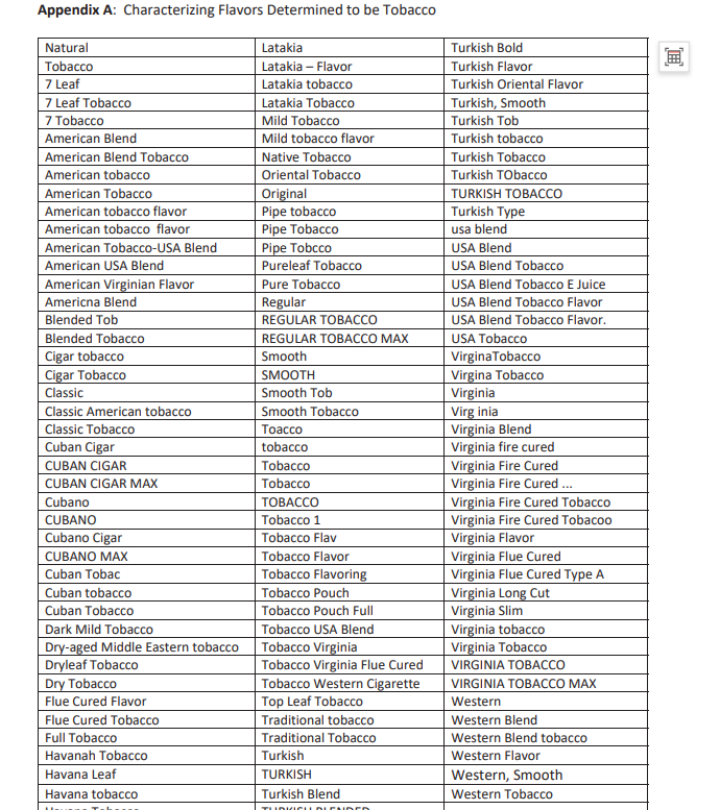
List of tobacco-flavored e-cigarette memos that have not undergone substantive scientific review | Source: FDA

List of tobacco-flavored e-cigarette memos that have not undergone substantive scientific review | Source: FDA

Attached list of tobacco-flavored e-cigarette memoranda that have not undergone substantive scientific review | Source: FDA
This is the fourth batch of memoranda issued in 2024, and the FDA has issued a total of 26 regulatory science policy memoranda. Although the FDA stated that the published memoranda should not be used as tools, guidelines or manuals for preparing applications or submitting documents to the FDA. However, the analysis and interpretation of these memoranda still help to have a deeper and more accurate understanding of the FDA's philosophy and methods for e-cigarette review.
The two supremes will continue to pay attention to the latest developments in the US e-cigarette regulatory policy.









