2Firsts Viewpoint | Technology for Good: The value, risks and solutions of flavored e-cigarettes in tobacco harm reduction, based on the latest scientific research and technology
Leave a message
2Firsts Viewpoint | Technology for Good: The value, risks and solutions of flavored e-cigarettes in tobacco harm reduction, based on the latest scientific research and technology
Flavored e-cigarettes have always been a controversial part of e-cigarette regulation. On the one hand, it is loved by consumers, but at the same time, it has also become the target of regulatory crackdowns due to its attraction to minors. A recent study released by ARAC, a well-known behavioral research organization in the United States, shows that flavored e-cigarettes have a significant effect on smoking cessation among adult tobacco consumers. However, this experiment did not cover the data of minors. The age verification technology recently released by Ispire may make up for this loophole. The continuous progress of scientific research and technology research and development will allow e-cigarettes to better return to their original intention of tobacco harm reduction. This not only brings the possibility to solve the problem of e-cigarette supervision, but is also conducive to the further development of the global NGP industry and THR industry.
Author: Echo Guo
Co-founder & COO of 2Firsts
Editor's note
ARAC (Applied Research and Analysis Corporation) provides illuminating evidence through a comprehensive study. At CORESTA 2024, ARAC presented results from its Longitudinal Randomized Experimental Switching Study (LRESS), which showed that flavored disposable e-cigarettes not only significantly reduced daily cigarette consumption, but also had higher smoking cessation rates than tobacco flavors Version.
At the 2024 CORESTA Conference, ARAC Chief Research Officer Dr. Jessica Zdinak presented the company's latest research results. As a leading global media and consulting company in the Novel Tobacco Products (NGP) industry, 2Firsts took a keen interest in these research results and collaborated with ARAC to obtain presentation PPTs. These include two studies - one focusing on nicotine pouches, which 2Firsts has previously published (click to view: Interpretation | Nicotine pouches are less attractive to non-smokers: The American behavioral agency ARAC under the FDA compliance framework study), another study on flavored e-cigarettes (ENDS) becomes the focus of this article. This article, written by 2Firsts, draws on key insights from the above speech PPT and ARAC research, as well as IKE Tech's recent report on flavored e-cigarette age verification technology, to explore potential solutions to the social problems caused by flavored e-cigarettes.
introduction
As the world's largest e-cigarette market, the United States accounts for about one-third of the global market share and leads international trends. In the past year or more, disposable e-cigarettes, which are mainly fruit-flavored, have attracted widespread social attention due to their appeal to minors. This issue has not only become the focus of media reports and regulatory discussions, but also highlighted the core issue of "flavoring" as an e-cigarette under the Appropriateness to Protect Public Health (APPH). Among them, the impact of flavored e-cigarettes on minors and their solutions are one of the most difficult and complex challenges in the industry.
Currently, only 34 electronic nicotine delivery system (ENDS) products have passed the FDA's PMTA review and obtained marketing authorization (MGO). These products are all limited to tobacco and menthol flavors. These approved products account for only 13.7% of the U.S. market share, and most of the market is still occupied by non-compliant flavoring products. Closing this gap will require not only stricter regulatory enforcement, but also advances in research and technology to address the complex intersection between flavored ENDS and accessibility to minors.
Addressing the social impact of flavored ENDS: Insights from LRESS and age verification technology
This ARAC study provides important insights into the effects of flavored electronic nicotine delivery systems (ENDS) on adult smokers, but leaves a significant gap in not exploring their effects on minors. Shortly after ARAC released its findings, Ispire Technology Inc. and IKE Tech LLC announced on November 18, 2024 that they had successfully completed a pre-PMTA submission with the U.S. Food and Drug Administration (FDA) Center for Tobacco Products (CTP) Meeting. The meeting focused on their age verification technology, which is designed to limit youth exposure to electronic nicotine delivery systems while using them, while ensuring adults have access to flavored products authorized through the PMTA.
Based on these developments, Dr. Zdinak previously shared his insights on the FDA's recent memorandum on PMTA applications for non-tobacco-flavored e-cigarettes when communicating with 2Firsts. She noted: "Age verification technology may help limit access to minors, but its effectiveness in reducing underage use is uncertain. Although various age verification technologies are emerging, the industry cannot assume that the introduction of these measures to avoid having to demonstrate a positive risk-benefit ratio in a PMTA application." Her views highlight the dual challenges faced by manufacturers: on the one hand, they need to integrate effective technical safeguards, and on the other hand, they need to provide adequate protection. Evidence that its products meet the Appropriateness to Protect Public Health (APPH) standards required for FDA approval.
Rigorous experimental design under the PMTA framework
The purpose of this study is to provide the FDA with longitudinal experimental data to evaluate whether candidate flavored disposable e-cigarette products meet the Appropriateness to Protect Public Health (APPH) standards. Specifically, the study aimed to determine whether these products could successfully promote smoking cessation or reduce cigarette consumption per day (CPD) by more than 50%. Additionally, studies have compared the effectiveness of flavored and tobacco-flavored e-cigarettes.
The study was conducted over six months in four regions across the United States, and participants were randomly assigned to two groups: one group used a combination of flavored and tobacco-flavored disposable e-cigarettes, and the other group used only tobacco-flavored disposable e-cigarettes. The experimental design was carefully constructed to take into account participants' smoking behavior, daily cigarette consumption, and success in quitting smoking. This method ensures the scientific rigor and reliability of the research results and provides solid data support for the FDA to review flavored disposable e-cigarette products under the PMTA framework.

*Participants randomly assigned to two groups in the LRESS study | Image source: ARAC
Six-month study: Flavored e-cigarettes outperform tobacco-flavored products in promoting smoking cessation
ARAC said this study was the first to use a rigorous experimental design to control for potential confounding variables, recruitment bias and sampling bias. The findings provide strong evidence that the effectiveness of cessation support for adult smokers in the United States increases significantly when adult smokers have a range of flavored and tobacco-flavored disposable e-cigarette products to choose from.
The first key finding from the study, which combined flavored and tobacco-flavor conditions with tobacco-flavor-only conditions, showed that exposure to disposable e-cigarette (ENDS) products significantly reduced smoking per day (CPD) at six months. Increase the smoking cessation rate during the experimental period.
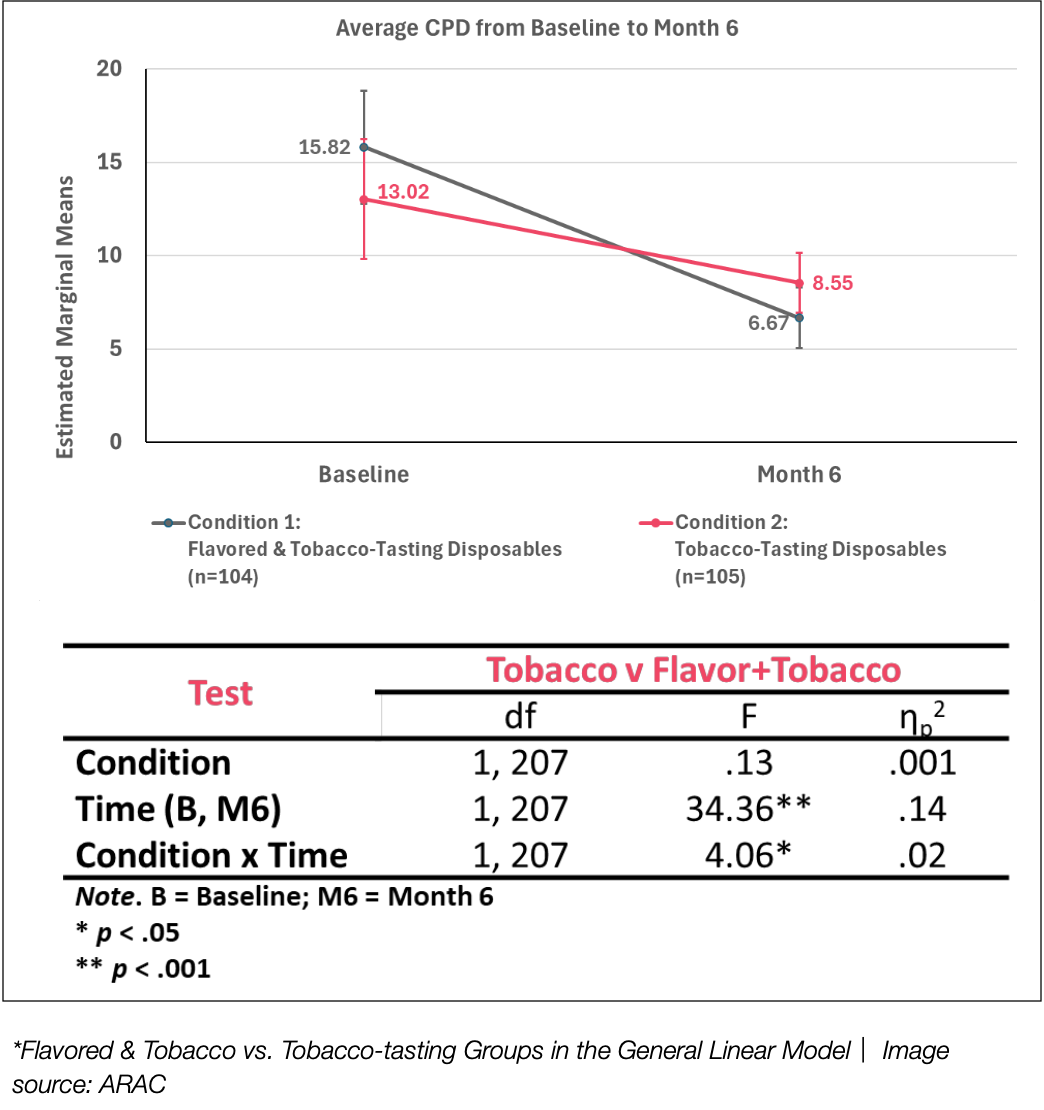
*Flavoured vs. tobacco group vs. tobacco flavor group in general linear model | Image source: ARAC
The second conclusion showed that although both groups experienced significant reductions in CPD, the group using flavored and tobacco-flavored disposable e-cigarettes had greater reductions after six months than the group using only tobacco-flavored e-cigarettes. Specifically, the CPD of condition 1 (flavored & tobacco flavor) dropped from 15.82 at baseline to 6.67, while condition 2 (tobacco flavor only) dropped from 13.02 to 8.55.
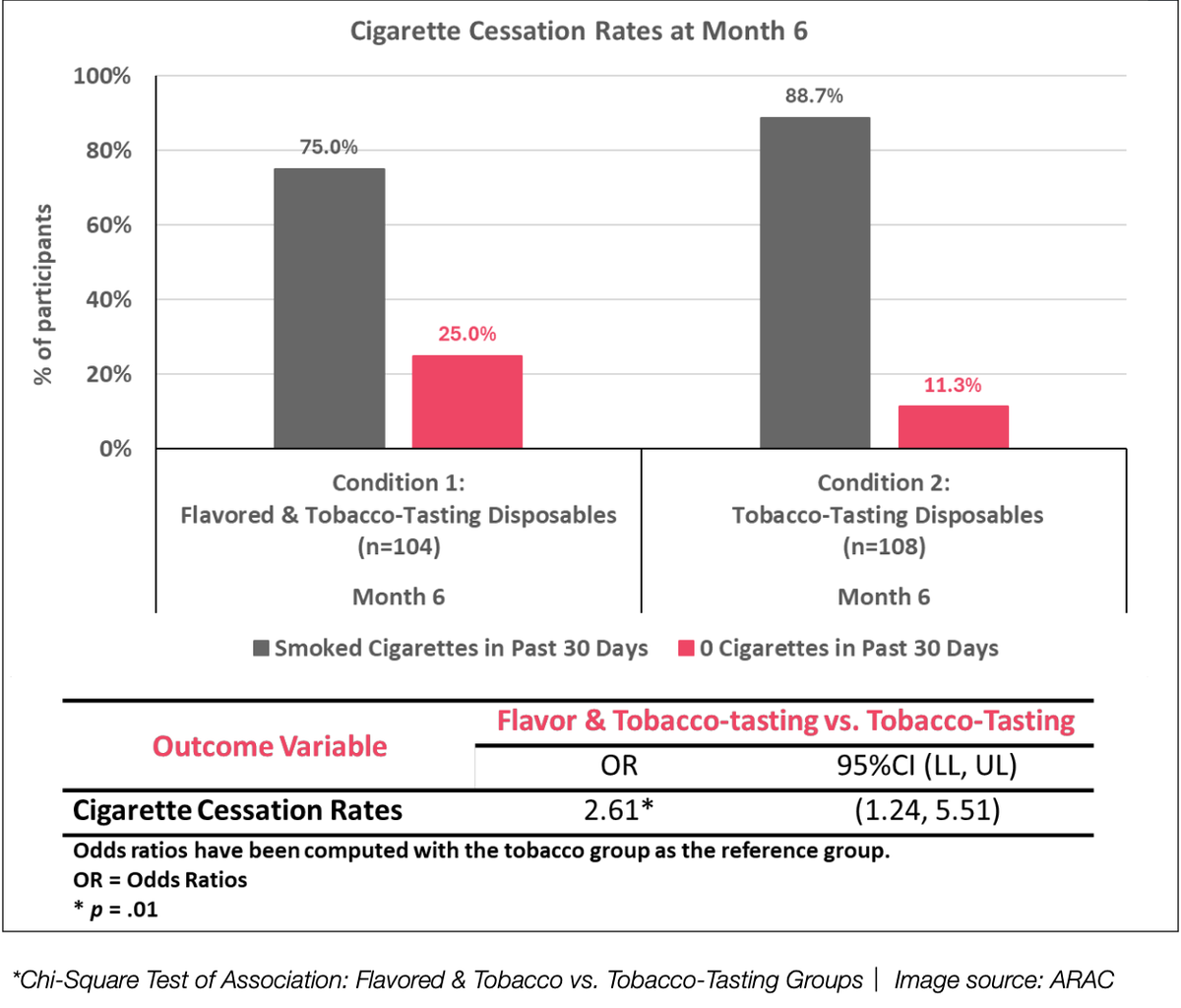
*Chi-squared association test: flavored and tobacco group vs. tobacco flavor group | Image source: ARAC
The third key finding showed that participants who used disposable e-cigarette products with flavors and tobacco flavors were 2.6 times more likely to quit smoking at the end of the six-month study than those who used disposable e-cigarette products containing only tobacco. This highlights the greater potential of flavored products for smoking cessation, further validating the greater reduction in daily smoking in the flavored group.
Addressing underage exposure: How age-verification technology is reshaping the debate on flavored ENDS
While ARAC is conducting research on APPH-related challenges of flavored ENDS, IKE Tech's age-verification system introduces a transformative technology solution. This innovation is particularly important because ARAC's recent LRESS did not explore youth exposure, and IKE Tech's technology is trying to fill this gap.
According to a joint statement from IKE Tech and Ispire, the system automatically initiates the age-verification process by pairing the ENDS device with the companion app via a Bluetooth connection. Users are required to provide valid identification, and the relevant information will be encrypted and stored in the blockchain ledger. The decentralized and immutable nature of blockchain enhances data security and authenticity, while the connection to a government-certified identity verification platform or authoritative database ensures that users meet the legal smoking age. Only verified adults can activate the device, effectively preventing minors from using ENDS.
IKE Tech plans to submit a PMTA for its age verification technology rather than a full PMTA for the finished product. This strategy is in line with FDA guidance that such submissions are acceptable as long as statutory requirements are met. IKE Tech said in its statement that the FDA may also grant priority review to the system due to its innovative use cases, thereby accelerating its integration into flavored ENDS products. This plug-and-play design is expected to simplify the compliance path and expand the approval potential for manufacturers.
To further promote technology adoption, IKE Tech plans to establish a Tobacco Product Master File (TPMF) for its components. Once authorized, the system and its TPMF will be available for manufacturers to license and integrate into their devices. This move is expected to promote compliance and innovation in the ENDS field and provide a solid framework for addressing regulatory challenges.
The FDA is stepping up enforcement against illegal flavored ENDS, signaling a clear direction of tighter regulation. At the same time, behavioral studies like ARAC's LRESS and technical innovations like IKE Tech's age verification system are accelerating the PMTA review process. These advances pave the way for the first flavored ENDS product to receive market authorization (MGO) through PMTA, a milestone that could reshape the entire industry.
This breakthrough will break the market structure that has long been dominated by non-compliant products and end the situation where bad money drives out good money. As the regulatory framework gradually improves, the competitive landscape is expected to change significantly, allowing compliant companies to become true market leaders. This change is expected to usher in a new era of greater sustainability and fairness for the next generation tobacco products (NGP) industry, driving the industry towards a direction that places equal emphasis on responsibility and innovation.









